| Identification |
| Name: | 8-Hydroxyquinoline |
| Synonyms: | 8-OQ; 8-oxyquinoline; 8-Quinolinol; Azanaphthalene-8-ol; fennosan; Fennosan H 30; HQ; hydroxybenzopyridine; Hydroxychinolin; Hydroxyquinoline; OQ; OXIN; Oxine; oxybenzopyridine; oxychinolin; oxyquinoline; phenopyridine; Quinolinol; quinophenol; Tumex; Quinolin-8-ol; Chinolin-8-ol; Quinoleine-8-ol |
| CAS: | 148-24-3 |
| EINECS: | 205-711-1 |
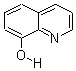
| Molecular Formula: | C9H7NO |
| Molecular Weight: | 145.16 |
| InChI: | InChI=1/C9H7NO/c11-8-5-1-3-7-4-2-6-10-9(7)8/h1-6,11H |
| Molecular Structure: |
 |
| Properties |
| Transport: | OTH |
| Density: | 1.03 |
| Stability: | Stable under normal temperatures and pressures. Darkens and decomposes on exposure to light. |
| Water Solubility: | INSOLUBLE |
| Solubility: | Insoluble in water |
| Appearance: | white to pale yellow crystal |
| Specification: |
?8-Hydroxyquinoline (CAS NO.148-24-3) is a white to off-white or faintly yellow crystalline powder with phenolic odor.It is?insoluble in water. 8-Hydroxyquinoline darkens on exposure to light and readily forms stable metal chelates. It is incompatible with strong oxidizers and it is also incompatible with many metal ions.
It? is a monoprotic bidentate chelating agent. Related ligands are the Schiff bases derived from salicylaldehyde, such as salicylaldoxime and salen. In neutral solution, the hydroxyl is in the protonated form (pKa=9.89) and the nitrogen is not protonated (pKa=5.13).However, an excited-state zwitterionic isomer exists in which H+ is transfered from the oxygen (giving an oxygen anion) to the nitrogen (giving a protonated nitrogen cation).
|
| HS Code: | 29334990 |
| Storage Temperature: | Store at RT. |
| Color: | White crystals or white crystalline powder |
| Usage: | Microbiocide, fungicide. |
| Safety Data |
| Hazard Symbols |
 Xn:Harmful
Xn:Harmful
|
| |
 |