| Identification |
| Name: | Tungsten fluoride(WF6), (OC-6-11)- |
| Synonyms: | Tungstenfluoride (WF6) (6CI,8CI); Hexafluorotungsten; Tungsten fluoride; Tungstenhexafluoride; Tungsten hexafluoride (WF6); Tungsten(6+) fluoride; Wolframhexafluoride |
| CAS: | 7783-82-6 |
| EINECS: | 232-029-1 |
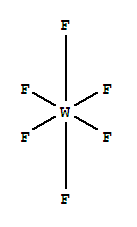
| Molecular Formula: | F6W |
| Molecular Weight: | 297.85 |
| InChI: | InChI=1/6FH.W/h6*1H;/q;;;;;;+6/p-6/rF6W/c1-7(2,3,4,5)6 |
| Molecular Structure: |
 |
| Properties |
| Transport: | UN 2196 2 |
| Melting Point: | 2 ºC |
| Flash Point: | °C |
| Boiling Point: | 17 ºC |
| Density: | 13.1 g/L, gas |
| Stability: | No data. |
| Water Solubility: | decomposes |
| Solubility: | Decomposes in water |
| Appearance: | Colorless gas |
| Specification: |
Tungsten(VI) fluoride, also known as tungsten hexafluoride, is the inorganic compound of tungsten and fluorine with the formula WF6. This corrosive, colorless compound is a gas under standard conditions. With a density of about 13 g/L (roughly 11 times heavier than air.), WF6 is one of the heaviest known gases under standard conditions.
|
| Report: |
Reported in EPA TSCA Inventory.
|
| Packinggroup: | O52 |
| Flash Point: | °C |
| Storage Temperature: | Keep in a cool, dry, dark location in a tightly sealed container or cylinder. Keep away from incompatible materials, ignition sources and untrained individuals. Secure and label area. Protect containers/cylinders from physical damage. |
| Usage: | Used in the manufacture of other chemicals and in the manufacture of electronics. |
| Safety Data |
| Hazard Symbols |
 T: Toxic
T: Toxic
 C: Corrosive
C: Corrosive
|
| |
 |